Posts Tagged ‘spectrochemical series’
Gee, I’m a Tree: Predicting the Geometry of Organometallic Complexes
An important issue that we’ve glossed over until now concerns what organometallic complexes actually look like: what are their typical geometries? Can we use any of the “bookkeeping metrics” we’ve explored so far to reliably predict geometry? The answer to the latter questions is a refreshing but qualified “yes.” In this post, we’ll explore the possibilities for complex geometry and develop some general guidelines for predicting geometry. In the process we’ll enlist the aid of a powerful theoretical ally, crystal field theory (CFT). CFT provides some intuitive explanations for geometry the geometry of OM complexes. Here we go!
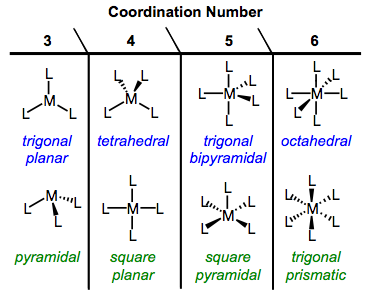
Because OM complexes feature a variety of coordination numbers, the possibilities for geometry are numerous. The common geometries of organic chemistry—linear, pyramidal, trigonal planar, and tetrahedral—are available to OM complexes too. Many complexes exhibit a second kind of four-coordinate geometry, square planar. Five-coordinate complexes may exhibit either square pyramidal or (my personal favorite) trigonal bipyramidal geometries. Six-coordinate complexes feature either octahedral geometry or the rare but intriguing trigonal prismatic arrangement. The figure below summarizes these possibilities (minus the two-coordinate geometries, which we won’t deal with).