Posts Tagged ‘acidity’
Epic Ligand Survey: σ Complexes
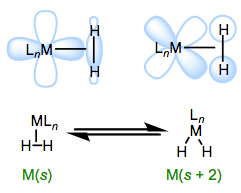
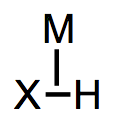 In this post, we’ll investigate ligands that, shockingly enough, bind through their σ electrons in an L-type fashion. This binding mode depends as much on the metal center as it does on the ligand itself—to see why, we need only recognize that σ complexes look like intermediates in concerted oxidative additions. With a slight reorganization of electrons and geometry, an L-type σ ligand can become two X-type ligands. Why, then, are σ complexes stable? How can we control the ratio of σ complex to X2 complex in a given situation? How does complexation of a σ bond change the ligand’s properties? We’ll address these questions and more in this post.
In this post, we’ll investigate ligands that, shockingly enough, bind through their σ electrons in an L-type fashion. This binding mode depends as much on the metal center as it does on the ligand itself—to see why, we need only recognize that σ complexes look like intermediates in concerted oxidative additions. With a slight reorganization of electrons and geometry, an L-type σ ligand can become two X-type ligands. Why, then, are σ complexes stable? How can we control the ratio of σ complex to X2 complex in a given situation? How does complexation of a σ bond change the ligand’s properties? We’ll address these questions and more in this post.
General Properties
The first thing to realize about σ complexes is that they are highly sensitive to steric bulk. Any old σ bond won’t do; hydrogen at one end of the binding bond or the other (or both) is necessary. The best studied σ complexes involve dihydrogen (H2), so let’s start there.
Mildly backbonding metals may bind dihydrogen “side on.” Like side-on binding in π complexes, there are two important orbital interactions at play here: σH–H→dσ and dπ→σ*H–H. Dihydrogen complexes can “tautomerize” to (H)2 isomers through oxidative addition of the H–H bond to the metal.
H2 binding in an L-type fashion massively acidifies the ligand—changes in pKa of over thirty units are known! Analogous acidifications of X–H bonds, which we touched on in a previous post, rarely exhibit ΔpKa > 5. What gives? What’s causing the different behavior of X–H and H–H ligands? The key is to consider the conjugate base of the ligand—in particular, how much it’s stabilized by a metal center relative to the corresponding free anion. The principle here is analogous to the famous dictum of organic chemistry: consider charged species when making acid/base comparisons. Stabilization of the unhindered anion H– by a metal is much greater than stabilization of larger, more electronegative anions like HO– and NH2– by a metal. As a result, it’s more favorable to remove a proton from metal-complexed H2 than from larger, more electronegative X–H ligands. Read the rest of this entry »
Epic Ligand Survey: Metal Hydrides
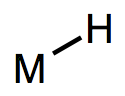 Metal-hydrogen bonds, also known (misleadingly) as metal hydrides, are ubiquitous X-type ligands in organometallic chemistry. There is much more than meets the eye to most M-H bonds: although they’re simple to draw, they vary enormously in polarization and pKa. They may be acidic or hydridic or both, depending on the nature of the metal center and the reaction conditions. In this post, we’ll develop some heuristics for predicting the behavior of M-H bonds and discuss their major modes of reactivity (acidity, radical reactions, migratory insertion, etc.). We’ll also touch on the most widely used synthetic methods to form metal hydrides.
Metal-hydrogen bonds, also known (misleadingly) as metal hydrides, are ubiquitous X-type ligands in organometallic chemistry. There is much more than meets the eye to most M-H bonds: although they’re simple to draw, they vary enormously in polarization and pKa. They may be acidic or hydridic or both, depending on the nature of the metal center and the reaction conditions. In this post, we’ll develop some heuristics for predicting the behavior of M-H bonds and discuss their major modes of reactivity (acidity, radical reactions, migratory insertion, etc.). We’ll also touch on the most widely used synthetic methods to form metal hydrides.
General Properties
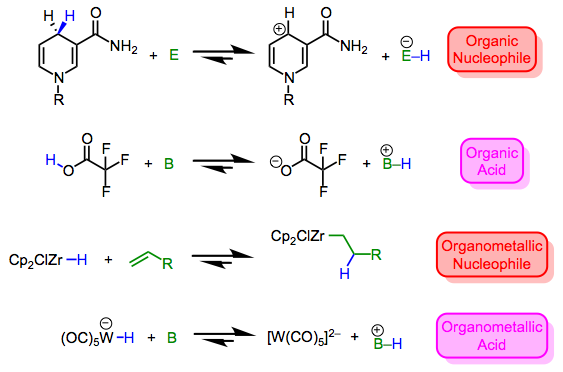
Metal hydrides run the gamut from nucleophilic/basic to electrophilic/acidic. Then again, the same can be said of X–H bonds in organic chemistry, which may vary from mildly nucleophilic (consider Hantzsch esters and NADH) to extremely electrophilic (consider triflic acid). As hydrogen is what it is in both cases, it’s clear that the nature of the X fragment—more specifically, the stability of the charged fragments X+ and X–—dictate the character of the X–H bond. Compare the four equilibria outlined below—the stabilities of the ions dictate the position of each equilibrium. By now we shouldn’t find it surprising that the highly π-acidic W(CO)5 fragment is good at stabilizing negative charge; for a similar reason, the ZrCp2Cl fragment can stabilize positive charge.*
Ligand Field Theory & Frontier Molecular Orbital Theory
In this post, we’ll begin to explore the molecular orbital theory of organometallic complexes. Some background in molecular orbital theory will be beneficial; an understanding of organic frontier molecular orbital theory is particularly helpful. Check out Fukui’s Nobel Prize lecture for an introduction to FMO theory. The theories described here try to address how the approach of ligands to a transition metal center modifies the electronics of the metal and ligands. The last post on geometry touched on these ideas a little, but we’ll dig a little deeper here. Notably, we need to address the often forgotten influence of the metal on the ligands—how might a metal modify the reactivity of organic ligands?
Ligand Field Theory
The ligand field theory (LFT) fleshes out the ideas of crystal field theory with molecular orbital theory concepts. It provides a method for understanding M–L bonding and antibonding orbitals; however, it has been strongly disputed by computational studies in favor of valence bond models that incorporate hypervalency. Still, LFT provides a more complete picture of complex bonding than crystal field theory, so we’ll discuss it here. Furthermore, the portions of LFT under dispute have nothing to do with CFT, so “no harm no foul.” Let’s take a look at the molecular orbitals of a hypothetical octahedral MLn complex to begin hashing out LFT.
Periodic Trends of the Transition Metals
Periodic trends play a huge role in organic chemistry. Regular changes in electronegativity, atomic size, ionization energy, and other variables across the periodic table allow us to make systematic predictions about the behavior of similar compounds. Of course, the same is true for organometallic complexes! With a firm grip on the periodic trends of the transition metals, we can begin to make comparisons between complexes we’re familiar with and those we’ve never seen before. Periodic trends essentially provide an exponential increase in predictive power. In this post, we’ll hit on the major periodic trends of the transition metals and discuss a few examples for which these trends can be handy.
Before beginning, a couple of caveats are in order. First of all, many of the trends across the transition series are not perfectly regular. Hartwig wisely advises that one should consider the transition series in blocks instead of as a whole when considering periodic trends. For instance, general increases in a quantity may be punctuated by sudden decreases; in such a case, we may say that the quantity increases generally, but definite conclusions are only possible when the metals under comparison are close to one another in the periodic table (and we need to be careful about unexpected jumps). Secondly, periodic trends are significantly affected by the identity of ligands and the oxidation state of the metal center, so comparisons need to be appropriately controlled. Using periodic trends to compare a Pd(II) complex and a Ru(III) complex is largely an exercise in futility, but comparing Pt(II) and Pd(II) complexes with similar ligand sets is reasonable. Keep these ideas in mind to avoid spinning your wheels unnecessarily! Alright, let’s dive in… Read the rest of this entry »