Posts Tagged ‘geometry’
Reductive Elimination: General Ideas
Reductive elimination is the microscopic reverse of oxidative addition. It is literally oxidative addition run in reverse—oxidative addition backwards in time! My favorite analogy for microscopic reversibility is the video game Braid, in which “resurrection is the microscopic reverse of death.” The player can reverse time to “undo” death; viewed from the forward direction, “undoing death” is better called “resurrection.” Chemically, reductive elimination and oxidative addition share the same reaction coordinate. The only difference between their reaction coordinate diagrams relates to what we call “reactants” and “products.” Thus, their mechanisms depend on one another, and trends in the speed and extent of oxidative additions correspond to opposite trends in reductive eliminations. In this post, we’ll address reductive elimination in a general sense, as we did for oxidative addition in a previous post.

A general reductive elimination. The oxidation state of the metal decreases by two units, and open coordination sites become available.
During reductive elimination, the electrons in the M–X bond head toward ligand Y, and the electrons in M–Y head to the metal. The eliminating ligands are always X-type! On the whole, the oxidation state of the metal decreases by two units, two new open coordination sites become available, and an X–Y bond forms. What does the change in oxidation state suggest about changes in electron density at the metal? As suggested by the name “reductive,” the metal gains electrons. The ligands lose electrons as the new X–Y bond cannot possibly be polarized to both X and Y, as the original M–X and M–Y bonds were. Using these ideas, you may already be thinking about reactivity trends in reductive elimination…hold that thought. Read the rest of this entry »
Gee, I’m a Tree: Predicting the Geometry of Organometallic Complexes
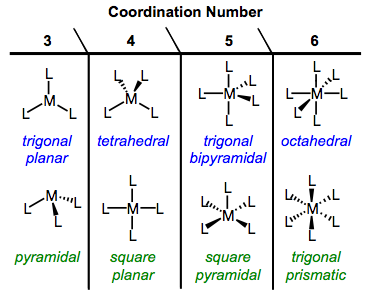
An important issue that we’ve glossed over until now concerns what organometallic complexes actually look like: what are their typical geometries? Can we use any of the “bookkeeping metrics” we’ve explored so far to reliably predict geometry? The answer to the latter questions is a refreshing but qualified “yes.” In this post, we’ll explore the possibilities for complex geometry and develop some general guidelines for predicting geometry. In the process we’ll enlist the aid of a powerful theoretical ally, crystal field theory (CFT). CFT provides some intuitive explanations for geometry the geometry of OM complexes. Here we go!
Because OM complexes feature a variety of coordination numbers, the possibilities for geometry are numerous. The common geometries of organic chemistry—linear, pyramidal, trigonal planar, and tetrahedral—are available to OM complexes too. Many complexes exhibit a second kind of four-coordinate geometry, square planar. Five-coordinate complexes may exhibit either square pyramidal or (my personal favorite) trigonal bipyramidal geometries. Six-coordinate complexes feature either octahedral geometry or the rare but intriguing trigonal prismatic arrangement. The figure below summarizes these possibilities (minus the two-coordinate geometries, which we won’t deal with).